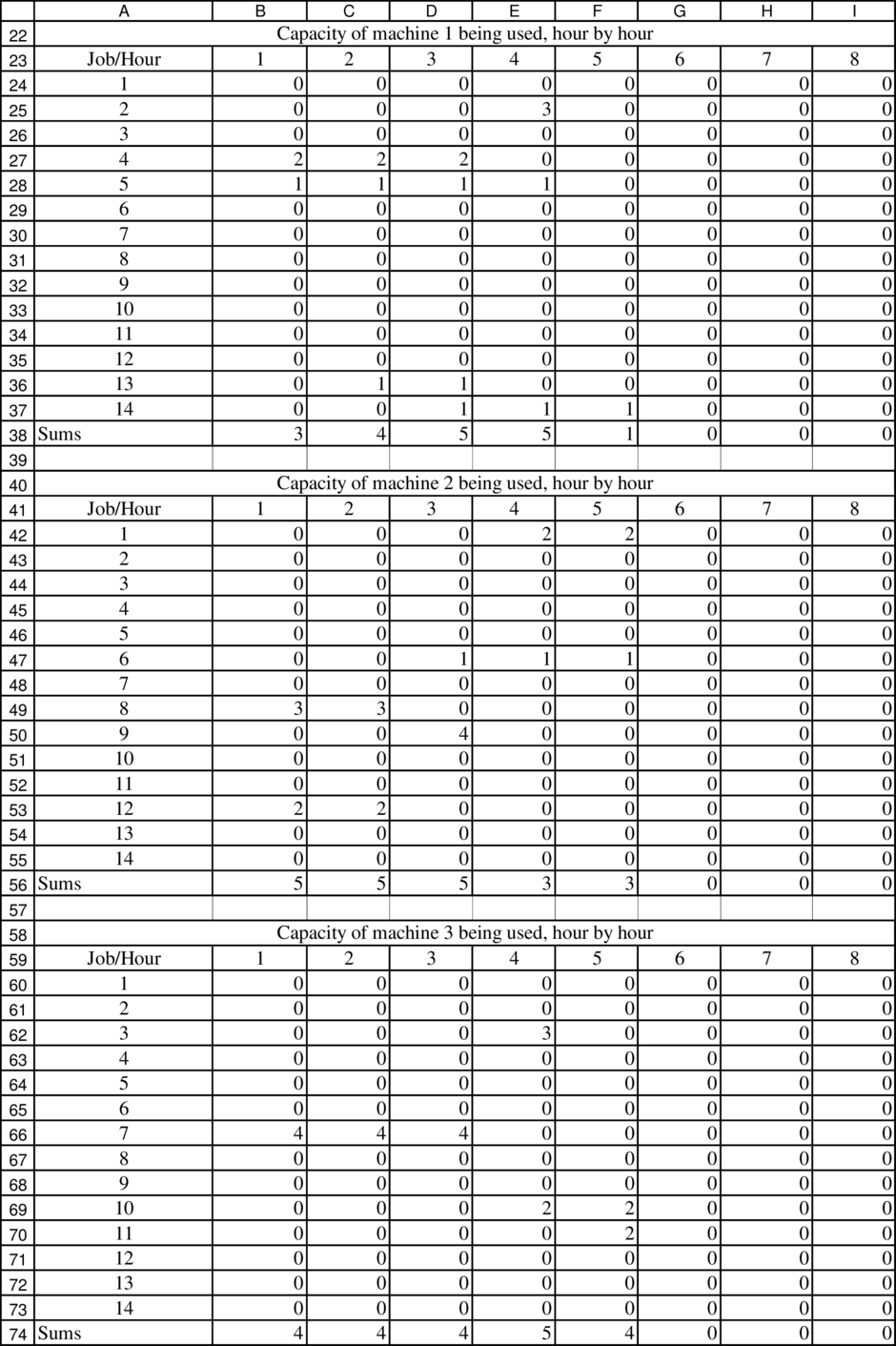
List of Electron Configurations of Elements.
Moving through the elements. in the periodic table, each atom has one more electron than the last because the number of electrons is the same as the atomic number. Electrons occupy the shells in.
The electron arrangement of sodium as a diagram Electron arrangements and the periodic table The electron arrangement of an element is related to its position on the periodic table.
The quickest way to write the electronic configuration of iodine, or any element, is to refer to its position in the Periodic Table. Iodine is a Group 17 (old IUPAC: Group VIIA) element, and all Group 17 elements have an outer electronic configuration of n s 2 n s 5, where n is the valence quantium shell corresponding to the period in which the element is in.
Read more by electron configuration and the periodic table. Electron configuration chart. Given below the sequence for filling electrons to the energy levels of an atom which is called the electron configuration chart. Electron Configuration Chart How to write the Electron configuration? There are three methods of writing the electron configuration. Orbital diagrams; spdf notation; Noble gas.
Complete Electron Configurations. To write a complete electron configuration for an uncharged atom, Determine the number of electrons in the atom from its atomic number. (See Below.) Add electrons to the sublevels in the correct order of filling. Add two electrons to each s sublevel, 6 to each p sublevel, 10 to each d sublevel, and 14 to each f sublevel. To check your complete electron.
The arrangement of elements in the Periodic Table is a result of the electron configuration of the atoms of the elements. Elements in a family (column) have similar physical and chemical properties because they have the same number and orbital type of valence electrons. For example, Alkali Metals Family (column 1) all have 1 s electrons. They differ in properties because they have different.
Electron Configuration Shortcut. Follow the steps below to write short cut version of electron configurations. Step 1: Find the element on the periodic table. Step 2: Go back to the last noble gas that was passed (atomic number). Step 3: Write the symbol of the noble gas in brackets to start your electron configuration. Put the atomic number of the noble gas beneath the symbol to let you know.